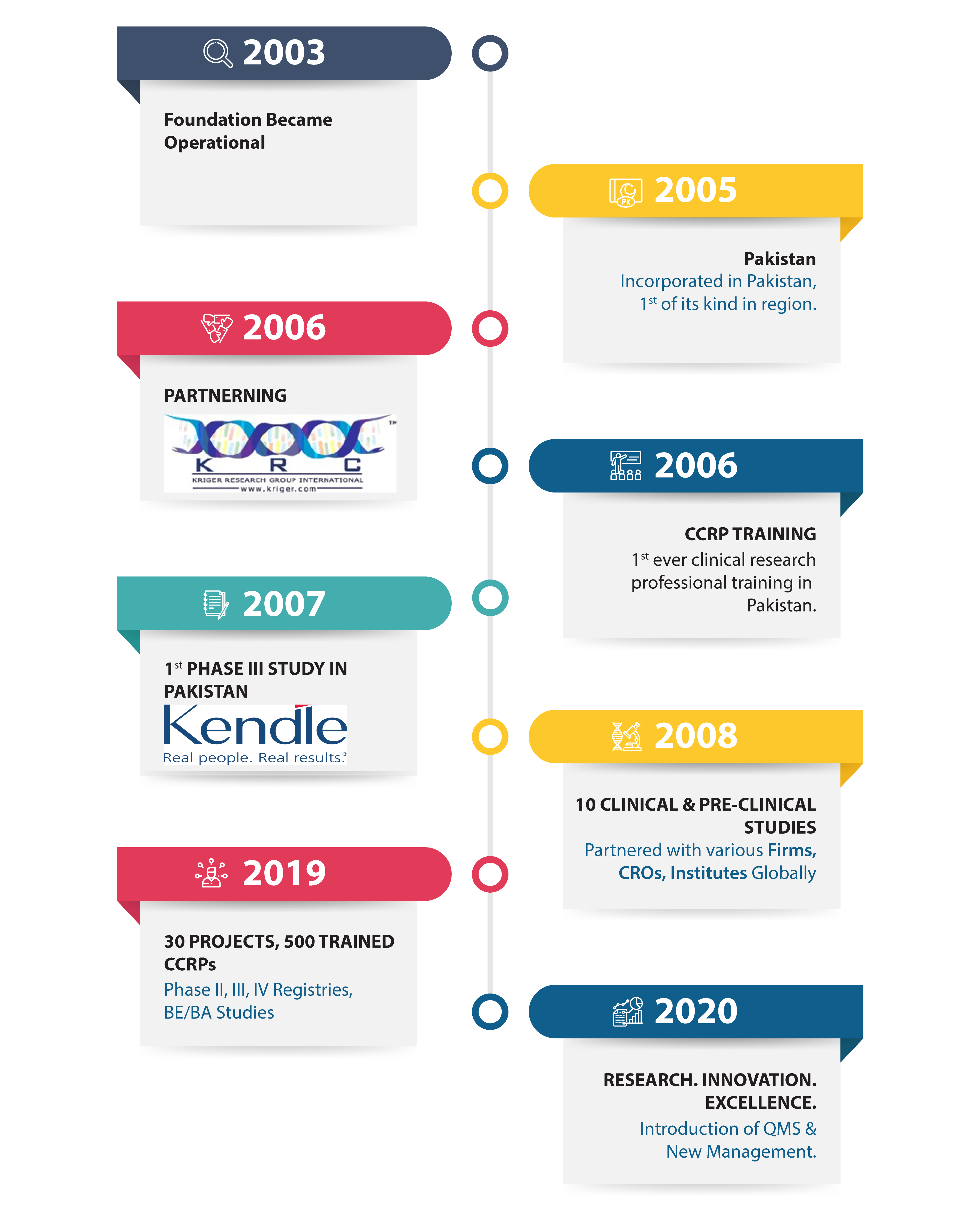
Metrics Research in 02 Decades
Our Collaborations
Some of The Many Pharmaceutical Companies
Some of The Many Globally Marked CROs
With Institutions in US, Europe, ASEAN and Pakistan

Our Mission
Our mission is to forge enduring partnerships with bio/pharmaceutical, medical device companies, CRO’s and institutions striving for healthcare excellence.
Our Vision
Our Vision is to become a leading CRO and clinical data science company that can help prevent and/or manage diseases and finding new cures.
QUALITY POLICY
Metrics Research is committed to providing the best possible services to all customers and other interested parties in terms of quality, integrity, efficiency, and value.
This commitment to quality extends from the management to all staff members and shall be regularly reviewed to ensure complete compliance with specified requirements.
This policy shall be supported by a fully documented Quality Management System based on the requirements of the International Standard ISO 9001:2015 to guarantee:
- Compliance with applicable regulations,
- Clients’ expectations and requirements regarding the services and clinical research trainings provided, and
- Continuous improvement of our activities.
Metrics Research recognizes the critical importance of Good Clinical Practice (GCP) and the need to guarantee patient’s rights and safety, as well as the reliability of data in clinical trials. In this regard, all our clinical operational activities are governed by strict adherence to ICH-GCP guidelines, local regulations, and appropriate quality standards to ensure the study’s success and customer satisfaction.
The policy and our Quality Management System will be regularly reviewed by management to ensure continued effectiveness and improvement through management reviews, training, and transparent communication to ensure a culture of continuous improvement throughout the company.

Approval: CEO
Metrics Research (Pvt) Ltd.
Date: 17th October 2022